A New Frontier in Peptide Therapy
One of the most exciting advances in the field of peptide therapy has been the introduction of a synthetic, cell-permeable peptide known as FOXO4-DRI that is designed to kill senescent cells.
First described in a landmark 2017 Cell paper, Baar et al.’s original report of this compound in the journal Cell in 2017 generated enormous interest and enthusiasm in the scientific and public communities. It posed an intriguing question:
Can a simple peptide reverse indicators of biological age in animals?
To address that question, it’s critical to understand what the mouse studies actually showed and what they didn’t and why that’s really important for the future of peptide therapy.
What Are Senescent Cells and Why Do They Matter?
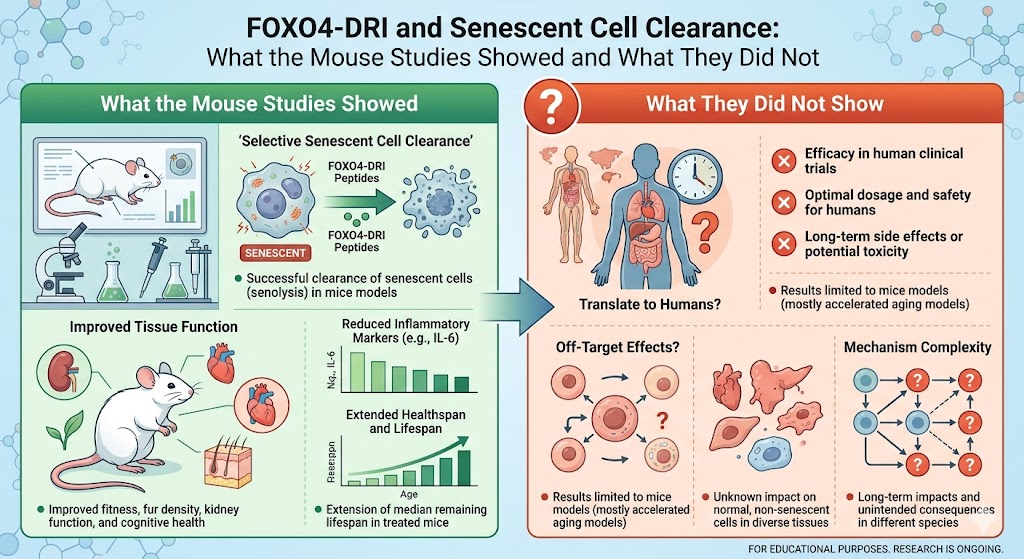
Senescence occurs when damaged or aged cells stop dividing but can’t die. Although this process was initially developed as an anti-tumor mechanism, recent research has shown that the prolonged accumulation of senescent cells drives inflammation, tissue degeneration, and disease in the elderly.
These cells release a toxic fireball of inflammatory factors called the senescence-associated secretory phenotype (SASP), which gradually impairs the function of their local environment.
Removing these cells, a process called “senolysis,” is therefore a prime research focus. The rationale is that if you silence the cell chatter, the tissue stands a chance of regaining its youthful vigor. This is the rationale behind FOXO4-DRI.
The Mechanism: More Than a Simple Peptide
FOXO4-DRI is no simple peptide. It’s a retro-inverso D-amino acid peptide , that is, a sequence of amino acids in the opposite order to their natural arrangement and with D-form (mirror image) amino acids, rather than the usual L-form amino acids. This property renders it resistant to enzymatic breakdown and able to cross cell membranes.
The way it works is clever: in senescent cells, the transcription factor FOXO4 binds p53 protein in an abnormal way, blocking the programmed cell death (apoptosis) that would normally clear senescent cells from the body. FOXO4-DRI displaces the FOXO4-p53 complex, allowing p53 to once again induce apoptosis
specifically in cells with high levels of FOXO4, such as senescent cells. In actively dividing cells, FOXO4 concentrations are too low to form the complex, so the peptide does not affect these cells. It is this selectivity that makes the proposed use of FOXO4-DRI in peptide therapy so interesting.
The Real Results: Here Is What Changed in Early Studies
Now, what happened when a little bit of FOXO4-DRI was injected into the biological systems of aged animals? So the initial results were encouraging, and they had implications in areas of interest to anyone who wants to know how peptide therapy can be used to promote physical function and well-being during the aging process.
There were some marked changes in physical function in the aged subjects. The subjects’ stamina, strength, and general activity improved. For anyone interested in peptides for muscle growth, or for those exploring ways to reverse age-related muscle loss, this was an important message . It indicated that removing senescent cells would have a positive impact on muscle regeneration in a way that restores physical performance that had been lost as we age.
It wasn’t all about muscle performance, either. Hair and skin regeneration also improved, especially when the accumulation of senescent cells was sped up. This highlights the wide-ranging effects a targeted peptide therapy can have on the entire tissue when it gets at the underlying problem: that is, dysfunctional cells.
There were also indications of improved kidney function. Healthy organs are related to the burden of senescent cells, and these data suggested that this approach may have broad effects. Most excitingly, though, healthy life expectancy seemed to increase, rather than mere life expectancy. That’s one of the key discussions we’re having in the field of peptide research: living longer vs. living better.
FOXO4-DRI Within the Broader Peptide Research Landscape
FOXO4-DRI is part of a rapidly evolving field of research. Other peptides currently being investigated (such as those investigated for growth, metabolic effects, and neurological function) are being investigated based on the same underlying assumption: that specific molecular signals can be used to restore or preserve cellular function.
A related field of increasing interest is sleep. The sleep-inducing peptide DSIP (Delta Sleep-Inducing Peptide) is another type of research chemical in which a single, bioactive sequence has been investigated for its role in regulating physiological processes.
in this case, the sleep cycle. Like FOXO4-DRI, DSIP research has yielded interesting preclinical results that remain to be carefully described in clinical studies. The point is this: good preclinical results are only the first step in research.
Promising Preclinical Data Deserves Rigorous Follow-Through
FOXO4-DRI is a scientifically and mechanistically plausible senolytic peptide approach. The animal studies demonstrated legitimate, consistent effects of selective senescent cell clearance and improved functional outcomes in senescent mice. That is no small achievement.
But the preclinical (mouse) data does not translate easily to clinical (human) applications. To properly engage with the FOXO4-DRI data, be it in research or teaching . We must hold both truths in our hands: the results are exciting, and they are also preliminary.
As peptide science advances, peers such as FOXO4-DRI (and indeed the growing corpus of sequenced peptides) will be subject to the same rigorous, data-driven considerations that are at the core of research into “the most exciting molecule on the planet.” Knowing what the data are (and are not) is the key to achieving this.
This is where the realization needs to be tempered by the reality. Recognizing the limitations of early work is not cause for despondency . It is the basis of sound, informed investigation.
The above results represent the very early phases of research. The journey from these early results to human use requires many more steps of research, safety testing, and clinical trials. It will take time, and it is time well spent.
The most interesting compounds in paramount peptide research have always been those that ask interesting questions and then are answered by good science. FOXO4-DRI is in that category.